Why the Japanese Market
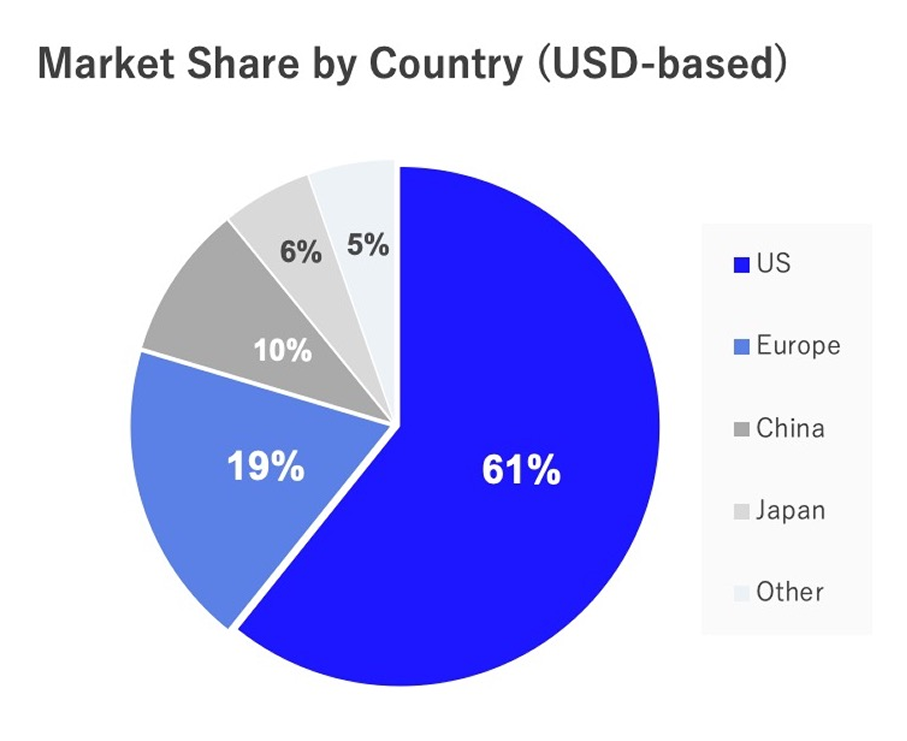
The World's 3rd Largest Market / Gateway to Asia
The World's 3rd Largest Market / Gateway to Asia
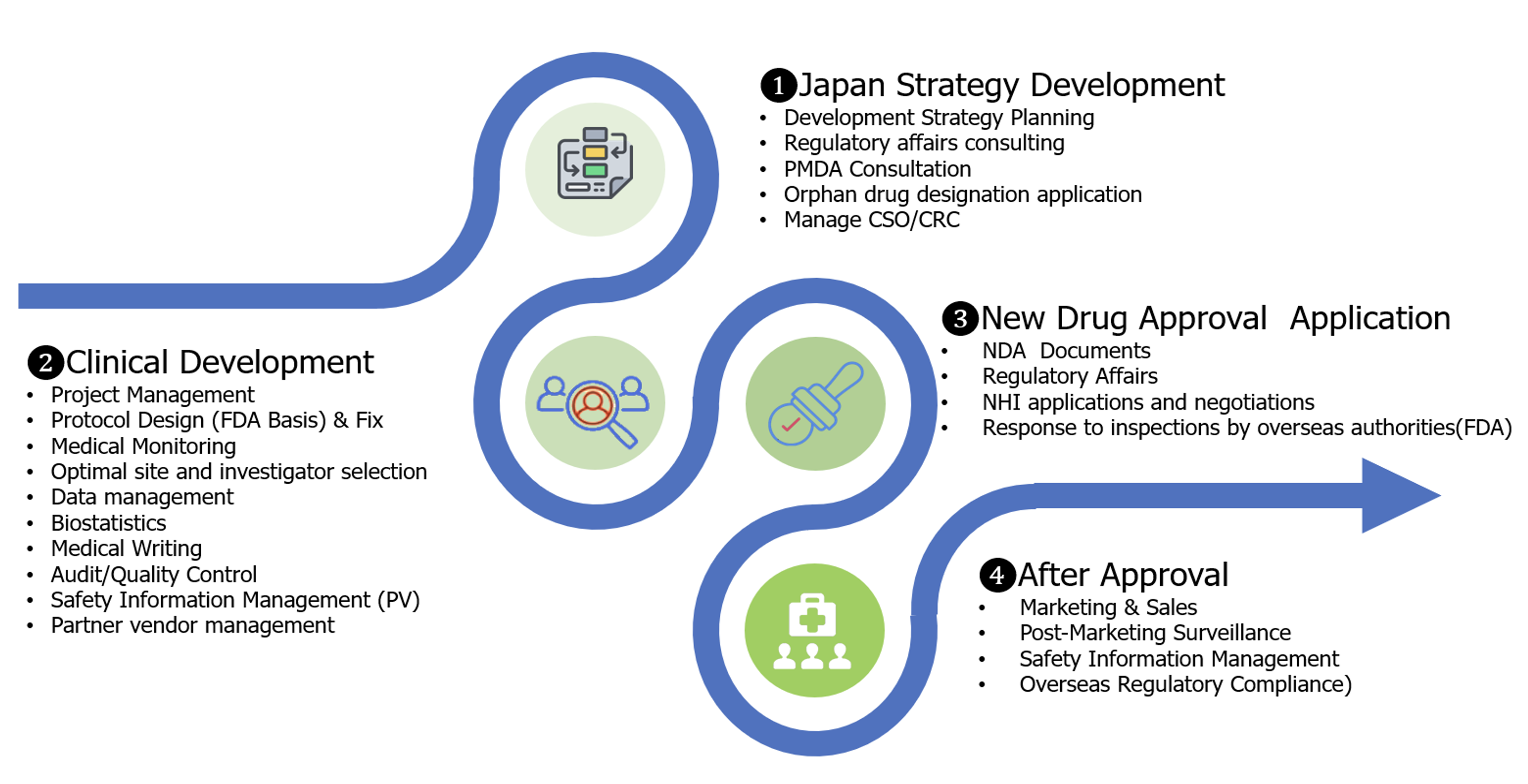
A Fast and Reliable Path to the Japanese Market
LinDo is committed to implementing the following three key solutions, to ensure your product achieves its maximum potential in the Japanese market, from market entry planning to market penetration.
Through alliance with our shareholder which has a panel of over 70% of Japanese physicians, we swiftly identify hospitals and clinical trial investigators who extensively treat the target disease for your product. This enables the quick identification of trial participation sites, allowing for efficient development that incorporates Japan into your global clinical trials.

Our team, well-versed in Japan's specific and complex regulatory and clinical development requirements, handles all process from trial design to negotiations with regulatory authorities. By accelerating patient enrollment in trials, we minimize the development period. Furthermore, we consistently manage the processes from regulatory application to pricing negotiations, securing optimal conditions to maximize your product's value. This ensures increased profitability after approval.
With the extensive experience of our members who have driven marketing and sales for global pharmaceutical companies in Japan, we accelerate market penetration. We can identify facilities and specialists who treat many patients suffering from the target disease based on proprietary data managed by our shareholder. We prioritize and deliver product information through a strategic mix of digital and conventional channels to these facilities and physicians, achieving early market establishment and sales expansion.

A Rapid and Reliable Path to the Japanese Market
As a specialty pharma company, LinDo provides a one-stop solution to these problems and will work with you on everything from the development to the sales of your products in Japan and the strengthening of your company brand.
LinDo is dedicated to maximizing your product's potential in the Japanese market.
If you are interested in collaborating with LinDo, we invite you to request a "Japanese Market Forecast Report,"
which will specifically outline the market opportunities for your product in Japan.
To create this report, our expert team will compile a valuable, tailored market forecast report for your product,
based on non-confidential information about your product and insights gathered from discussions with your team.
Please feel free to contact LinDo to arrange a discussion with our specialized team.